Introduction
Numerous molding techniques are well-known for mass-producing commonplace items. Still, a number of businesses require more complex parts and components to be produced with conventional plastic injection molds (Jyo, 2023). High-precision injection molding of plastic is the process used to meet such manufacturing demands. This effective injection molding method uses accurate molds and is a highly advanced technology. The fact that it is used to generate more complex plastic pieces rather than full items makes it more accessible and effective to meet the diverse needs of current plastic goods production. Healthcare relies heavily on medical injection molding because it makes it possible to quickly and effectively create intricate, compatible parts for a wide range of medical instruments. Syringes, tubes, implants used for surgery, and medication delivery devices are a few examples of them. It’s a really practical and affordable solution. But in order to fulfill the growing demand for medical products, perfect accuracy must coexist with mass manufacture. To ensure the efficiency, security, and dependability of these crucial parts, this accuracy is necessary. This post will go over the key techniques for producing medical injection molding at a fast and accurate pace (tronicarts – Multimedia-Agentur, 2016).
Challenges of balancing speed and precision
In production, accuracy and speed frequently clash. Increased output through faster cycle times is not without its obstacles. The operation of medical devices may be compromised by molded components that have surface defects, shrinkage, and warping as a result of faster injection rates and shorter cooling periods. Quicker cycles also translate into less time spent adjusting process parameters, which might lead to a deviation from the requirements(Injection Molding Specialists | Same-Day Quote | Aberdeen Tech., n.d.).
Precision and speed balance are particular issues in medical injection molding. Tight regulatory constraints necessitate materials of a medical grade that satisfy particular standards. Due to their higher melt viscosity than typical plastics, these materials need to have their parameters adjusted for optimal flow and to prevent flaws. Medical components with critical dimensions require precision molding with tight tolerances.
Strategies of high speed and precision production
Several strategies can be implemented.
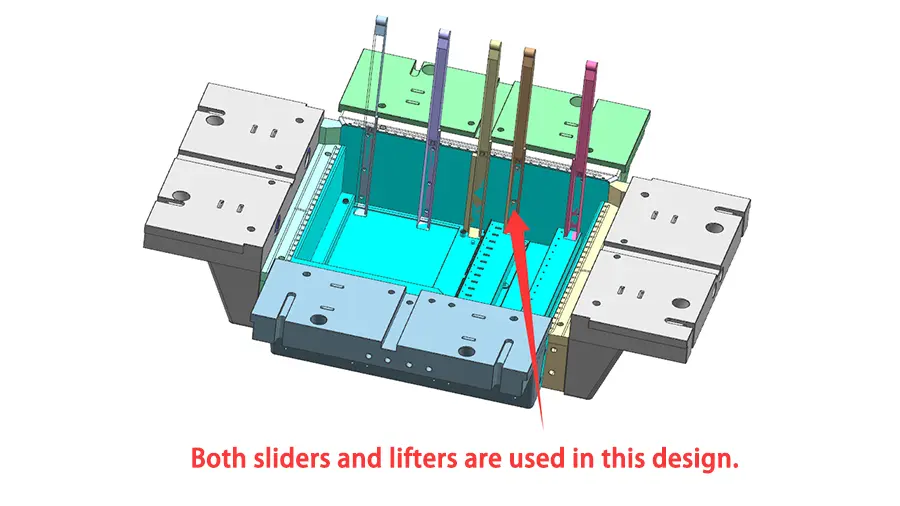
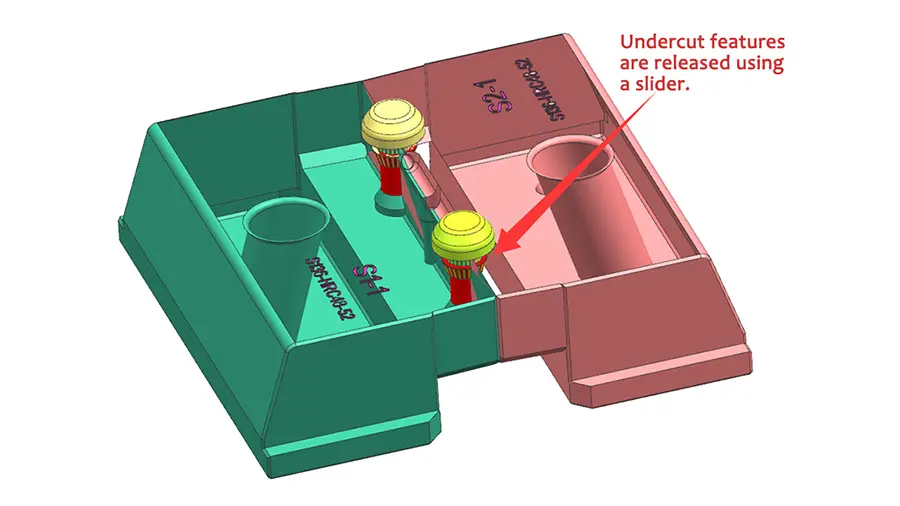
1. Mold design and manufacture
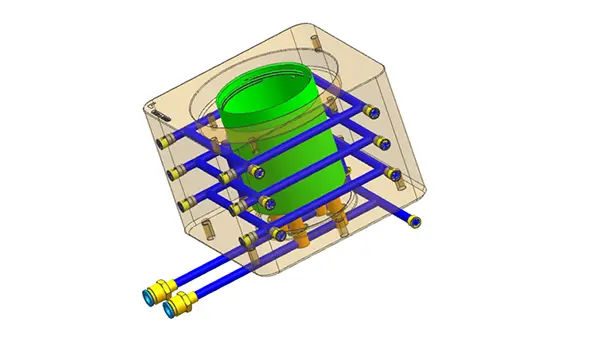
The design and construction of the mold itself provide the framework for an effective molding process. To reduce warping and preserve uniform part dimensions, premium mold steels with outstanding dimensional stability are essential. In order to achieve consistent filling and reduce stress spots on the molded object, methods such as carefully placing runners and gates are part of the process of improving mold design for effective material flow. Furthermore, adding features like adherent cooling pathways can shorten cooling periods considerably, resulting in quicker cycle times without sacrificing component quality(Peixoto et al., 2022).
Acquiring speed and accuracy is further enhanced by the use of sophisticated mold production procedures. Multiple pieces may be produced in a single cycle thanks to multi-cavity molds, which greatly boosts output. Hot runner systems save cycle times and save material waste by doing away with the need to remove sprue and runners. Last but not least, using precise machining and polishing procedures guarantees the molded components have an excellent surface quality, which is essential for some medical applications.
2. Material selection and optimization
It is crucial to choose the right medical-grade material to guarantee processability, performance, and biocompatibility. Melt flow index and thermal conductivity are two material parameters that affect the mold’s cooling and flow characteristics. These qualities are maximized by using the right drying and preheating methods, which also minimize shrinkage in the finished components and provide steady flow. Moreover, investigating cutting-edge materials for particular uses, such as bioresorbable polymers, might provide extra advantages including regulated bodily breakdown(Gülçür et al., 2021).
3. Process control and monitoring
Consistent component quality in high-speed manufacturing requires a strong quality management system to be put in place. Real-time data on crucial parameters like as injection pressure, melt temperature, gripping force, mold temperature, cycle duration, and cooling rates may be obtained by sophisticated process monitoring systems. This data may be analyzed using statistical process control methods to find any changes in the process that can cause faults. Manufacturers can guarantee the quality of every molded item and maintain constant process parameters by implementing corrective measures based on the data.
Advanced technologies for high speed and precision
Modern medical the use of injection molding manufacturing heavily relies on automation and robots. Part removal and material handling automation speeds up production and simplifies the procedure. The integration of in-mold decoration and secondary processing procedures may streamline the molding process and expedite production schedules by obviating the need for separate stages. Additionally, consistent component dimensions and surface quality may be guaranteed at large production volumes using automated quality control inspections utilizing vision systems (RAPID DIRECT, 2022).
Beyond traditional injection molding methods, cutting-edge technologies provide ways to produce complicated medical components even more quickly and precisely. For certain medical devices, multi-shot injection molding makes it possible to form pieces with many colors or materials in a single cycle. Micro-injection molding serves the need for producing very precise and small-scale medical components with precise dimensions. Lastly, before actual manufacturing starts, sophisticated simulation tools may be used to improve mold design, material selection, and process parameters. Virtual prototyping speeds up development times and increases first-shot success rates by drastically reducing trial-and-error procedures (Peixoto et al., 2022).
Maintaining sterility and regulatory compliance
When producing medical devices, maintaining sterility is crucial. Manufacturing free from contamination is ensured by stringent mold cleaning and validation procedures. For medical equipment, sustaining cleanliness throughout the manufacturing process is essential. To avoid contaminating the molded components, proper mold cleaning and validation protocols are necessary. Environments with cleanrooms are essential for reducing the possibility of contaminants. Strict adherence to regulatory standards, such as ISO 13485, ensures thorough documentation and strong quality control, both of which are necessary for regulatory compliance.