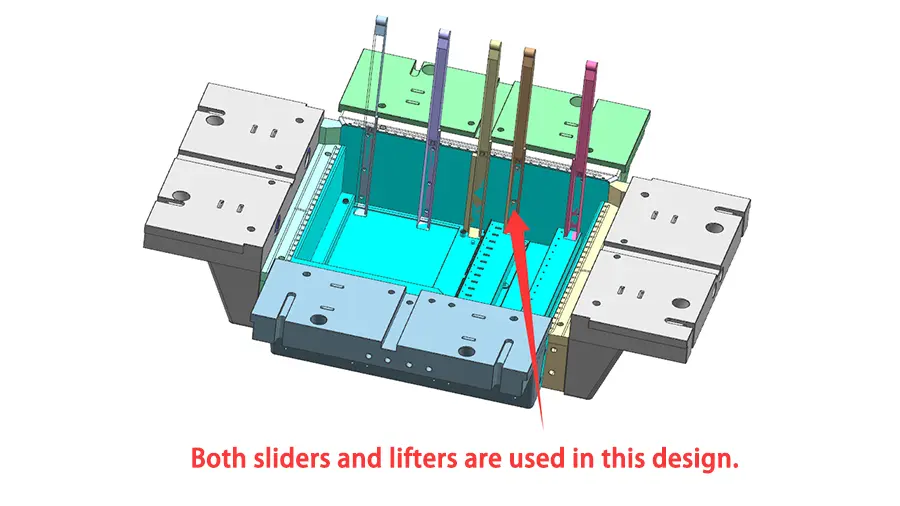
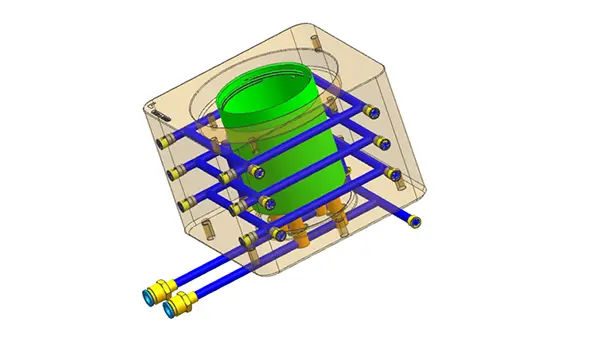
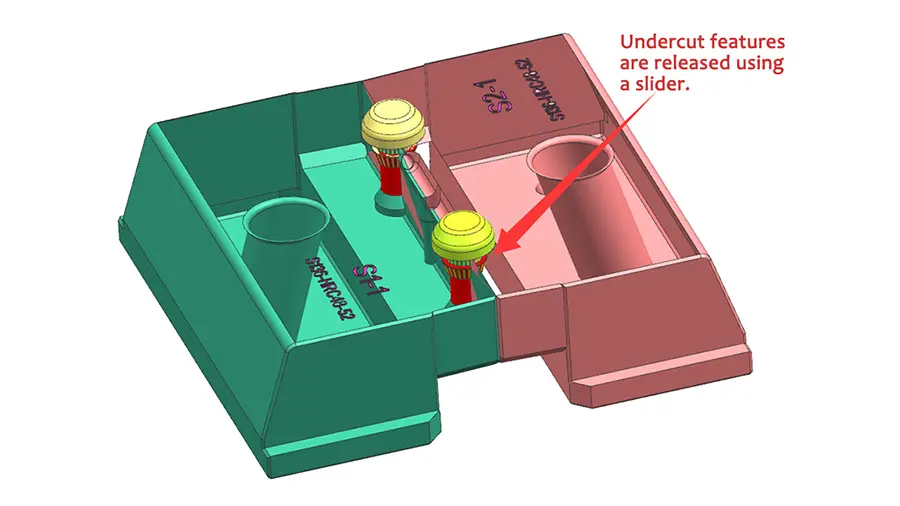
Injection molding is a widely used manufacturing process in the medical industry, providing many advantages for a variety of applications. Medical grade plastic injection molding is used for medical devices, components, laboratories and facilities. One of the key advantages of the injection molding process is its ability to meet the stringent regulatory and regulatory needs of the medical industry, which we discuss in further detail below.

Benefits of injection molding for medical devices
Medical suppliers have found that plastic injection molding medical parts, components and equipment is one of the most efficient ways to obtain the required quality and specifications in an economical and efficient manner. Plastic injection molding for medical devices is used in the following applications:
- Buildable component
- Beakers, test tubes and other containers
- Enclosures and enclosures for medical and laboratory equipment
- Surgical equipment and components
- Administration equipment and components
- The orthopaedic
These are just a few ways to use plastic injection molding for medical devices. The process of plastic injection molding medical parts can be applied to almost any field requiring mass production of durable, sterilization-friendly parts with high production accuracy.
When utilizing injection molding, medical industry suppliers and manufacturers can reap multiple benefits inherent in the process and available materials. These benefits include:
- Cost effectiveness – The nature of injection molding makes it one of the more cost effective manufacturing processes, especially in high volume production. Injection molding is almost always the most cost-effective option when mass-producing medical parts, components and equipment.
- Superior precision – Precision tolerance production is critical in medical device manufacturing and millimetres or microns can affect the successful performance of components. Skilled plastic injection molding equipment can produce a large number of components and equipment with great precision, and the differences between parts are negligible.
- Material selection – Plastic injection molding has one of the widest selection of materials of any manufacturing process. While medical-grade requirements will certainly narrow this range, the amount of material available to meet these critical specifications is still significant. We will discuss material selection later in this article.
- Durability – Many plastics used in injection molding have excellent strength and durability and can resist harsh environments, vibration and blunt force without cracking, breaking or shattering. Many injection plastics are also heat resistant and can be repeatedly sterilized by autoclave and other means.
- Pollution resistance – Plastic materials used in injection molding of medical devices are designed to be resistant to contamination and easy to sterilize. Moreover, the injection molding process itself can easily meet FDA and other requirements.
Notes on injection molding of medical devices

Plastic injection molding for medical devices is a critical process in which the risk of failure is very high. Therefore, several factors must be considered in the design, planning and manufacturing process. These include:
- FDA Regulations — As noted above, FDA’s cleanliness and sterility regulations are the primary consideration for medical components, both in the component itself and during manufacturing. Ensure that the materials you choose meet these standards and that your manufacturing partners can pass audits or other regulatory requirements to obtain medical grade approval.
ISO Certification and Compliance – ISO regulations govern processes and outputs for a wide range of industries, including the healthcare industry. At a minimum, facilities manufacturing medical components must comply with ISO 13485:2022 and further meet class I, II or III requirements, depending on the product being manufactured. Biocompatibility standards,
- Material properties – Materials selected for medical components and devices must have specific physical properties to be suitable for these applications. These include:
- Disinfection capability – This is a minimum requirement for any medical component, whether it is an equipment casing in a facility or part of a device that interacts directly with the human body. The material must be resistant to contamination and sterilized to prevent injury to those who come in contact with it.
- Suitability for Extreme Environments — Biological implants must be able to cope with the harsh environment of the human body, including reliable operation and durability under exposure to heat, fluids, corrosion, vibration and other movements. Fortunately, many plastics used for injection molding meet these requirements.
- Durability — Fragile materials are not practical in the medical industry because they are dangerous and inconvenient. Ensure that the material you choose has the fragmentation resistance required in the medical field.
- Selection of injection materials for medical devices
Now that you understand the benefits of plastic injection molding for medical components and devices and the specific requirements that those components must meet, we’ll look at some specific materials that meet the requirements. In general, plastics are superior to other materials and processes in terms of design flexibility, overall cost-effectiveness and production speed.

Consider the following materials for your project:
- Silicone – Silicone is often the material of choice for components and parts where flexibility is a key requirement. It has excellent durability and biocompatibility, and is very cost-effective in mass production.
- Polycarbonate – this is one of the strongest materials, with high impact and vibration resistance. Polycarbonate is ideal for high-visibility components because it can be made almost completely transparent. It also has strong heat resistance and uv resistance.
- Polyethylene — Polyethylene has a high molecular weight and is ideal for use as a surface for prosthetics and wearables due to its appearance, durability and relative smoothness. It is high strength and cost effective, but cannot be sterilized by autoclave.
- Polypropylene — Due to its high heat resistance, this material is ideal for components that must be repeatedly sterilized by autoclave. It also shows remarkable radiation resistance.With these skills, you will be able to make informed decisions about the materials and processes that best suit your medical device or component manufacturing.

RJC was established in 2002 and engaged in engineering service and technical manufacturing, such as rapid prototyping, mold manufacturing, injection molding, and CNC machining.CNC machining.RJC would be a good vendor choice.
click :https://rjcmold.com/contact-us/
Send inqury.